Therapeutic Modulation of Ubiquitin Ligase Activity
Lenalidomide, and other immunomodulatory drugs (IMiDs), have powerful clinical activity in del(5q) myelodysplastic syndrome (MDS) and multiple myeloma (MM), but the mechanism of drug activity was previously unknown. We demonstrated that lenalidomide induces the degradation of specific protein substrates that are essential for the expansion of MM and MDS cells. We are exploring novel ways to hijack the IMiD paradigm, screening other essential cancer genes, and testing drugs to target them in preclinical and clinical trial.
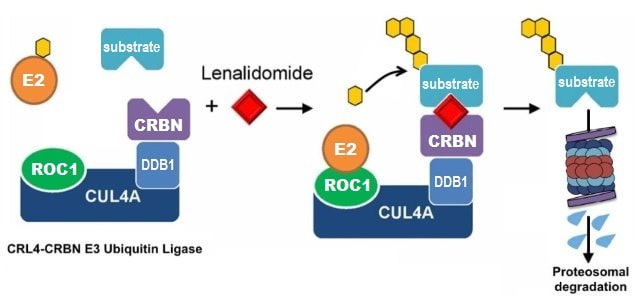
Lenalidomide, a derivative of thalidomide, has powerful clinical activity in del(5q) myelodysplastic syndrome (MDS) and multiple myeloma (MM), but the mechanism of drug activity was previously unknown. We demonstrated that thalidomide derivatives induce the degradation of specific proteins by modulating the function of the CRL4-CRBN E3 ubiquitin ligase. Degradation of IKZF1 and IKZF3 are critical for the therapeutic activity of these drugs in multiple myeloma, and degradation of CK1α is critical for the activity of lenalidomide in del(5q) MDS. These and other compounds can induce degradation of additional proteins, creating the potential for additional biological and clinical activity.
Lenalidomide, a derivative of thalidomide, has powerful clinical activity in del(5q) myelodysplastic syndrome (MDS) and multiple myeloma (MM), but the mechanism of drug activity was previously unknown. We demonstrated that thalidomide derivatives induce the degradation of specific proteins by modulating the function of the CRL4-CRBN E3 ubiquitin ligase. Degradation of IKZF1 and IKZF3 are critical for the therapeutic activity of these drugs in multiple myeloma, and degradation of CK1α is critical for the activity of lenalidomide in del(5q) MDS. These and other compounds can induce degradation of additional proteins, creating the potential for additional biological and clinical activity.